Milija Sarajlić
Current Research
Simple manufacturing of micro-sized Au thin film based Mercury sensor
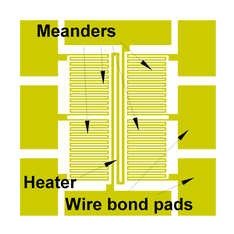
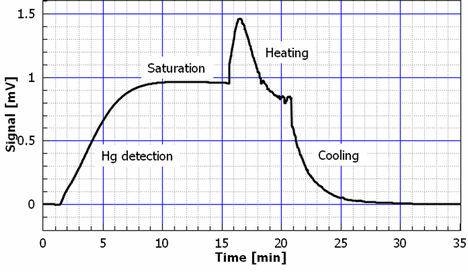
Mercury contamination presents renewed threat to the European Countries due to the newly enhanced production of different types Discharge Lamps instead of incandescent light bulbs [1]. This comes as a addition to the already existing sources of mercury in environment from battery industry, coal fired power plants, dental amalgams and gold purification. We decided to push development of Au thin film based mercury sensor as a part of national program for environmental protection. Two goals are important: simplified and affordable technology for mass production as well as sensitivity, portability and miniaturization. Here we report on fabrication details, sensitivity testing and measurement application cycling. Sensor is based on Thin Gold Film resistivity change within Wheatstone bridge configuration [2]. We fabricated miniaturized sensor in respect to the previously reported developments [3]. Also, technology is simplified comparable to the other authors [4]. On the both surfaces of double side polished Silicon wafer, 2 µm thick SiO2 is formed by thermal oxidation. Subsequently, on one of the surfaces Au thin film is deposited by sputtering technology, thickness 150 nm. As underlayer for gold film, chromium is used, 10 nm thick. Au thin film is patterned in a single lithography step with photoresist spin coating, mask exposure, photoresist development and Au-Cr thin film pattern formation. Fig 1 gives shape of the fabricated Au-Cr film. Gold film thickness of 150 nm proved to be thick enough for wire bonding on the sensor pads, Fig 2, at the same time providing thin enough film for mercury vapor sensing. This trade-off comes as advantage of simplified technology process. Smallest features at the sensor surface are meander conductive lines having width of 20 µm and separation of 20 µm as well. Total die size is 3.2x3.2 mm.Two diagonal meanders are covered by photoresist applied in a single step of spin coating, followed by mask exposure, development and removal. In this way, other two diagonal meanders are exposed to the metallic mercury from vapor phase in the Wheatstone bridge configuration providing increased sensitivity to the resistivity change. Fig 3 gives complete cycle of measurement and sensor restoration. Sensor is temporarily exposed to the 10 mg/m3 mercury concentration. Operating current is 1 mA, input voltage 0.43 V, maximum output bias is close to 1mV. Wheatstone bridge signal is swiftly increased reaching saturation after five minutes. After saturation level is reached, sensor is brought to the atmosphere of clean N2, but apparently signal is fixed at the saturation value. Heater on the surface is applied to evaporate adsorbed Hg on meanders. Signal is shortly increased due to the temperature elevation but promptly starts to fall as the mercury is desorbed from Au surface. After certain bias is reached, heating is turned off and signal comes to the starting point. Thus one measurement cycle is completed. Langmuir time dependent mass of adsorption is given in Eq 1, [5] where mHg is the mass of adsorbed mercury, mHgsat is the saturation Hg mass, t is the time, tau is the characteristic time of adsorption. Output signal during adsorption on Fig 3 gives dependence that resembles Eq 1 by which we may conclude that mass of adsorbed Hg to the meander resistivity has linear dependence. |
(1) |
[1] European Commission Directorate-General Environment Contract: ENV.G.2/ETU/2007/0021, Options for reducing mercury use in products and applications, and the fate of mercury already circulating in society, FINAL REPORT December 2008. [2] John J. McNerney, Peter R. Buseck, Roland C. Hanson, Science, New Series, Vol. 178, No. 4061 (Nov. 10, 1972), pp. 611-612. [3] B. Mazzolai, V. Mattoli, V. Raffa, G. Tripoli, D. Accoto, A. Menciassi, P. Dario, Sensors and Actuators A 113 (2004) 282–287. [4] Kwan Schambach, Entwurf, Herstellung und Charakterisierung eines mikromechanischen Quecksilbersensors, Ph.D. Thesis, Dortmund University, 2006, pp 13. [5] I. Langmuir, J. Am. Chem. Soc. 40 (1918). S. 1361